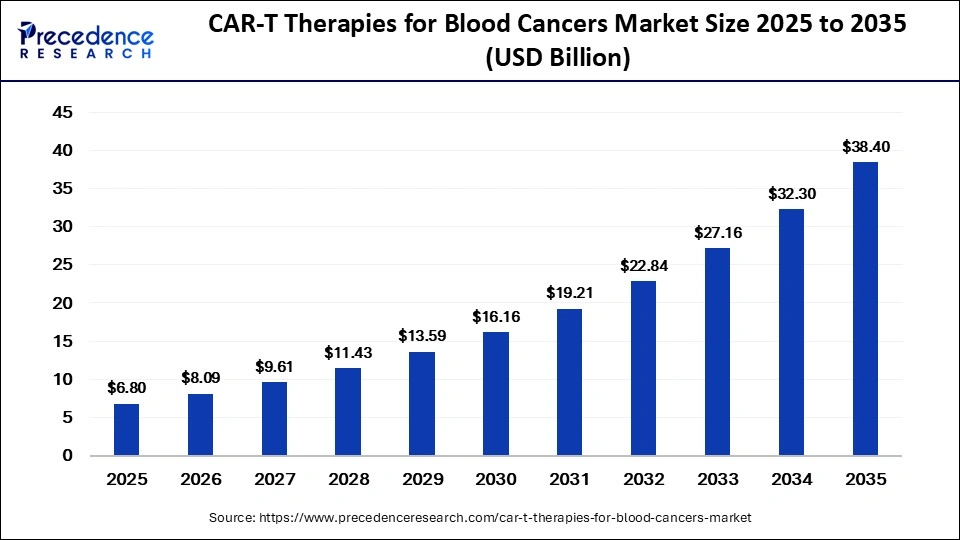
The global CAR-T therapies for blood cancers market is entering a transformative growth phase, with its valuation expected to rise from USD 8.09 billion in 2026 to approximately USD 38.40 billion by 2035, expanding at a robust CAGR of 18.90%. This rapid expansion is largely fueled by the clinical success of CAR-T cell therapies in treating relapsed and refractory blood cancers, alongside a surging demand for personalized and targeted oncology treatments.

As healthcare systems worldwide pivot toward precision medicine, CAR-T therapies are redefining treatment paradigms for leukemia, lymphoma, and multiple myeloma offering long-term remission possibilities where conventional therapies often fall short.
CAR-T Therapies for Blood Cancers Market Key Insights
- The market was valued at USD 6.80 billion in 2025 and is projected to reach USD 38.40 billion by 2035, reflecting strong double-digit growth.
- North America led the global market with a dominant 50% share in 2025 due to advanced healthcare infrastructure and early adoption.
- Asia Pacific is emerging as the fastest-growing region, expected to expand at a CAGR of 22.5% during the forecast period.
- CD19-targeted CAR-T therapies dominated the market with a 45% share in 2025 owing to proven efficacy in B-cell malignancies.
- Autologous CAR-T therapies accounted for 80% of the market share in 2025, driven by established clinical success and regulatory approvals.
- Hospitals remained the leading end-users, contributing 50% of total revenue due to specialized treatment capabilities.
How is AI Transforming the CAR-T Therapies Market?
Artificial intelligence is becoming a critical enabler in the evolution of CAR-T therapies. By analyzing vast genomic and clinical datasets, AI accelerates the identification of tumor-specific antigens, significantly improving the precision of CAR design. This reduces development timelines and enhances therapeutic success rates.
Moreover, AI-powered machine learning models are optimizing patient selection by predicting which individuals are most likely to benefit from CAR-T therapy. In manufacturing, AI is streamlining T-cell engineering, improving quality control, and ensuring scalability—ultimately reducing production complexities and costs.
CAR-T Therapies for Blood Cancers Market Key Growth Drivers
- Rising Demand for Personalized Medicine: CAR-T therapies represent a paradigm shift from generalized chemotherapy to individualized treatment. By engineering patient-specific T-cells, these therapies deliver higher response rates and durable outcomes, especially in relapsed cancer cases.
- Increasing Cancer Burden: The global rise in hematologic malignancies, including leukemia and lymphoma, is significantly boosting demand for advanced therapies like CAR-T.
- Strong R&D Investments: Biopharmaceutical companies are heavily investing in next-generation CAR-T technologies, including gene editing and multi-target approaches, accelerating innovation pipelines.
- Regulatory Support & Approvals: Expanding FDA approvals for new indications and earlier-line treatments are improving accessibility and adoption globally.
CAR-T Therapies for Blood Cancers Market Opportunities and Trends
- Donor-derived therapies eliminate the need for patient-specific manufacturing, enabling faster treatment delivery and scalability. Their cost-effectiveness and accessibility are expected to transform global adoption.
- Dual-target and multi-antigen CAR-T therapies are reducing relapse rates by preventing antigen escape, significantly improving treatment durability.
- Innovations like CRISPR-based editing are improving precision, reducing toxicity, and enabling the development of safer next-generation therapies.
CAR-T Therapies for Blood Cancers Market Key Metrics
| Report Coverage | Details |
| Market Size in 2025 | USD 6.80 Billion |
| Market Size in 2026 | USD 8.09 Billion |
| Market Size by 2035 | USD 38.49 Billion |
| Market Growth Rate from 2026 to 2035 | CAGR of 18.90% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2025 |
| Forecast Period | 2026 to 2035 |
| Segments Covered | Target Antigen, Cancer Type, Therapy Type, End-Use, Manufacturing Type, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
CAR-T Therapies for Blood Cancers Market Segmentation
By Target Antigen
| Segment | 2025 Share | 2035 Share | CAGR |
|---|---|---|---|
| CD19-targeted | 45% | 40% | 15.5% |
| BCMA-targeted | 30% | 35% | 21.5% |
| CD20-targeted | 10% | 9% | 14.5% |
| Dual/Multi-antigen | 15% | 16% | 22% |
CD19 therapies dominate due to strong clinical validation, while BCMA therapies are rapidly gaining traction in multiple myeloma. Multi-antigen therapies are emerging as the fastest-growing segment.
By Cancer Type
| Cancer Type | 2025 Share | CAGR |
|---|---|---|
| DLBCL | 35% | 16.5% |
| Multiple Myeloma | 30% | 22% |
| ALL | 20% | 15% |
| CLL | 10% | 14.5% |
Diffuse large B-cell lymphoma leads due to early adoption, while multiple myeloma is the fastest-growing segment driven by strong pipeline innovations.
By Therapy Type
- Autologous CAR-T therapies dominated with an 80% share in 2025 due to established clinical use.
- Allogeneic therapies are expected to grow at a 25% CAGR, offering scalability and faster delivery.
By End-Use
- Hospitals led with a 50% share due to advanced infrastructure and expertise.
- Specialty cancer centers are rapidly expanding, growing at a CAGR of 20.5%.
By Manufacturing Type
- In-house manufacturing held 60% share due to quality control advantages.
- Contract manufacturing (CDMOs) is growing at 21.5% CAGR, driven by outsourcing trends and scalability needs.
CAR-T Therapies for Blood Cancers Market Regional Insights
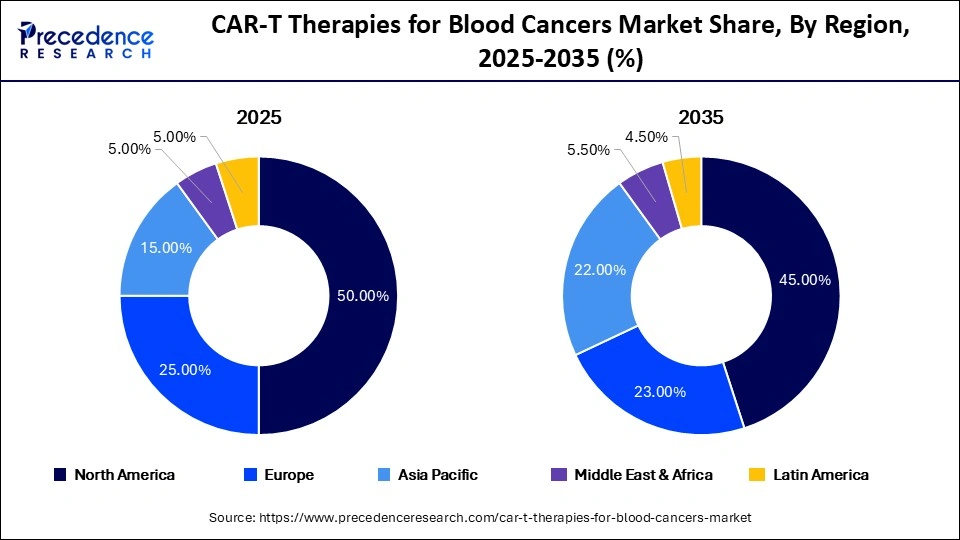
North America accounted for 50% of the market in 2025, supported by strong R&D, leading biotech companies, and advanced healthcare systems. The U.S. remains the innovation hub with rapid regulatory approvals and funding support.
Asia Pacific is projected to grow at the fastest CAGR of 22.5%, driven by rising cancer incidence, expanding healthcare infrastructure, and strong government support in countries like China and India.
Europe holds a 25% market share, benefiting from favorable reimbursement policies, increasing treatment centers, and strong clinical research initiatives.
CAR-T Therapies for Blood Cancers Market Leading Companies
- Novartis AG
- Gilead Sciences, Inc. (Kite Pharma)
- Bristol Myers Squibb Company
- Johnson & Johnson (Legend Biotech)
- Fate Therapeutics, Inc.
- Allogene Therapeutics, Inc.
- Cellectis S.A.
- Bluebird Bio, Inc.
- CRISPR Therapeutics AG
- Autolus Therapeutics plc
Latest Breakthroughs and Developments
- Bristol Myers Squibb launched Breyanzi for marginal zone lymphoma, expanding CAR-T indications.
- Johnson & Johnson received European approval for Darzalex combination therapy in multiple myeloma.
- AdAlta and SHcell partnered to develop CAR-T therapy for solid tumors, marking expansion beyond blood cancers.
Segments Covered in the Report
By Target Antigen
- CD19-targeted CAR-T Therapies
- BCMA-targeted CAR-T Therapies
- CD20-targeted CAR-T Therapies
- Dual-target & Multi-antigen CAR-T Therapies
By Cancer Type
- Acute Lymphoblastic Leukemia (ALL)
- Diffuse Large B-Cell Lymphoma (DLBCL)
- Multiple Myeloma
- Chronic Lymphocytic Leukemia (CLL)
- Other Hematologic Malignancies
By Therapy Type
- Autologous CAR-T Therapies
- Allogeneic (Off-the-shelf) CAR-T Therapies
By End-Use
- Hospitals
- Specialty Cancer Treatment Centers
- Academic & Research Institutes
By Manufacturing Type
- In-house Manufacturing
- Contract Manufacturing (CDMO-based)
By Region
- North America
- Latin America
- Europe
- Asia-pacific
- Middle and East Africa
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/8377
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344
