The global oncology landscape is undergoing a major transformation as in vivo CAR-T therapy emerges as one of the most promising innovations in cancer immunotherapy. Unlike conventional ex vivo CAR-T treatments that require extraction and engineering of immune cells outside the patient’s body, in vivo CAR-T therapy directly reprograms T cells internally using advanced gene delivery technologies.
The global in vivo CAR-T therapy market was valued at USD 650 million in 2025 and is projected to grow from USD 861.25 million in 2026 to approximately USD 10,841.07 million by 2035, expanding at an extraordinary CAGR of 32.50% from 2026 to 2035. The market is being driven by the increasing prevalence of cancer, rising clinical trial activity, advancements in gene-editing technologies, and growing investment in next-generation immunotherapies.
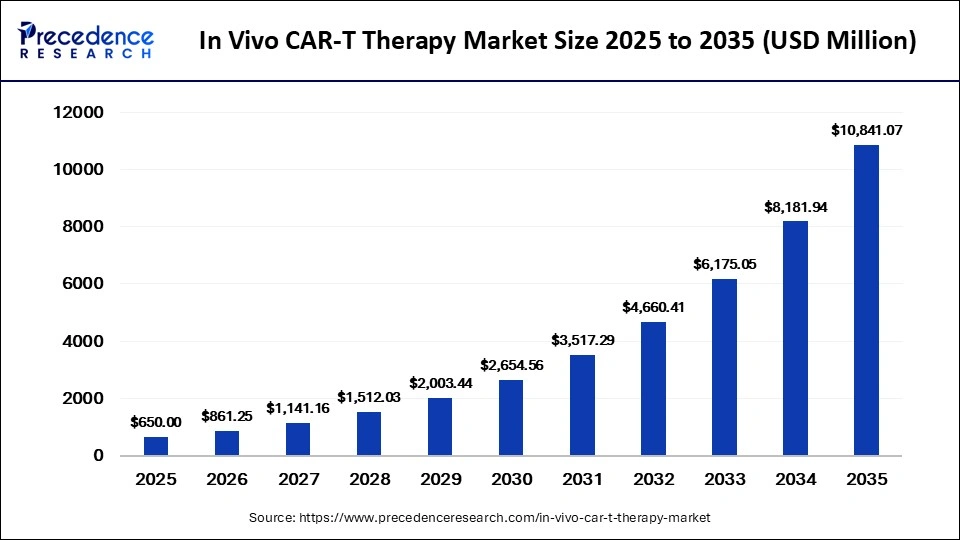
As healthcare systems seek faster, scalable, and cost-efficient alternatives to traditional CAR-T manufacturing processes, in vivo CAR-T platforms are rapidly attracting attention from pharmaceutical companies, research institutions, and investors worldwide.
Read Also: Large-Scale Active Stem Cell Clinical Trials Market
Quick Insights: What Are the Key Market Highlights?
North America dominated the market with a 50% revenue share in 2025 due to advanced biotechnology ecosystems and strong clinical research infrastructure.
Asia Pacific is projected to witness the fastest growth during the forecast period owing to increasing clinical trials and expanding biotech investments.
The viral vector-based segment accounted for 65% of the market share in 2025 because of its strong gene delivery efficiency and early clinical adoption.
The CD19 target antigen segment held a dominant 35% share due to its success in treating B-cell malignancies.
Hematologic malignancies represented 60% of total application demand in 2025, making oncology the largest therapeutic area.
Systemic delivery methods accounted for 70% market share due to ease of administration and broader treatment applicability.
Hospitals dominated the end-use segment with a 45% share because of their advanced oncology treatment infrastructure.
What Is In Vivo CAR-T Therapy and Why Is It Transforming Oncology?
In vivo CAR-T therapy is an advanced immunotherapy approach that genetically programs T cells directly inside a patient’s body using viral vectors, lipid nanoparticles, or other delivery platforms. This approach eliminates the need for complex ex vivo manufacturing procedures such as cell extraction, laboratory engineering, and reinfusion.
The therapy has demonstrated strong potential for treating hematologic malignancies such as leukemia, lymphoma, and multiple myeloma while also opening new opportunities in solid tumors and autoimmune diseases. By simplifying manufacturing and reducing treatment timelines, in vivo CAR-T therapy could significantly improve accessibility and scalability in cancer care.
Researchers believe this technology could reshape the future of personalized oncology by delivering faster, more affordable, and highly targeted immunotherapies.
AI Role in the Market: How Is Artificial Intelligence Accelerating In Vivo CAR-T Development?
Artificial intelligence is playing a transformative role in the advancement of in vivo CAR-T therapy. AI-powered systems are helping researchers optimize CAR construct design, improve targeted delivery precision, and predict treatment outcomes with greater accuracy.
Machine learning algorithms are also enabling the discovery of novel tumor antigens, analyzing tumor heterogeneity, and enhancing safety modeling to reduce off-target immune responses. These technologies are accelerating therapy development timelines while improving clinical trial efficiency.
AI-driven strategies are additionally being used to enhance CAR manufacturing processes and improve delivery vehicle engineering. As in vivo CAR-T therapies become increasingly data-intensive, AI is expected to become central to precision immunotherapy innovation.
Market Growth Factors: What Is Driving Market Expansion?
Why Is Rising Cancer Prevalence Increasing Demand for In Vivo CAR-T Therapy?
The growing global burden of hematologic malignancies and solid tumors is increasing demand for highly targeted immunotherapies capable of delivering long-term treatment outcomes.
How Are Clinical Trials Accelerating Market Growth?
The increasing number of ongoing preclinical and clinical-stage in vivo CAR-T therapy programs is strengthening the market pipeline and attracting major biotechnology investments.
Why Is Cost Efficiency Creating New Opportunities?
In vivo CAR-T therapy has the potential to significantly reduce manufacturing complexity, shorten turnaround times, and improve patient convenience compared to conventional ex vivo CAR-T processes.
How Are Advanced Gene Delivery Platforms Supporting Innovation?
Technologies such as viral vectors, mRNA lipid nanoparticles, circular RNA systems, polymeric nanoparticles, and CRISPR-based integration platforms are improving safety, scalability, and delivery precision.
Opportunities & Trends: What’s Shaping the Future of the Market?
Are Non-Viral Delivery Platforms Becoming the Next Big Opportunity?
Yes, non-viral vector systems are projected to grow at the fastest rate because they offer lower immunogenicity, improved scalability, and enhanced design flexibility.
Can In Vivo CAR-T Therapy Expand Beyond Blood Cancers?
Researchers are increasingly exploring solid tumor targets such as HER2, EGFR, and mesothelin, expanding CAR-T applications beyond hematologic cancers.
Is Localized Delivery Emerging as a High-Growth Segment?
Localized delivery methods are gaining momentum because they improve drug concentration at tumor sites while reducing systemic toxicity and side effects.
How Are Next-Generation CAR-T Cells Improving Therapeutic Outcomes?
Third-generation CAR-T cells incorporating multiple signaling domains are improving T-cell persistence, functionality, and effectiveness in resistant tumor environments.
Segmentation Analysis: Where Are the Key Revenue Streams?
By Vector Type
- Viral vector-based therapies: 65% share
- Non-viral vector-based therapies: fastest-growing segment due to lower immunogenicity and greater scalability
By Target Antigen
- CD19: 35% share
- BCMA: rapidly growing due to multiple myeloma applications
- Solid tumor targets: fastest-growing segment driven by expanding oncology research
By Application
- Hematologic malignancies: 60% share
- Solid tumors: fastest-growing application segment
- Autoimmune diseases: emerging treatment area with strong future potential
By Delivery Method
- Systemic delivery: 70% share
- Localized delivery: expected to grow at the fastest CAGR through 2035
By End-Use
- Hospitals: 45% share
- Specialized cancer treatment centers: fastest-growing segment
- Academic & research institutes: critical innovation hubs
Regional Analysis: Which Regions Are Leading the Market?
Why Does North America Dominate the Market?
North America leads the market due to its strong biotechnology ecosystem, advanced healthcare infrastructure, active clinical pipeline, and supportive FDA regulatory framework. The U.S. remains the largest contributor to regional market growth.
How Strong Is Europe’s Market Position?
Europe maintains a strong position because of rising academic collaborations, regulatory advancements, and increasing investment in advanced immunotherapy research. Germany is emerging as a key innovation hub within the region.
Why Is Asia Pacific the Fastest-Growing Region?
Asia Pacific is witnessing rapid growth due to increasing cancer prevalence, supportive government initiatives, rising biotechnology investments, and expanding clinical research infrastructure in China, Japan, and other emerging markets.
Competitive Landscape: Who Are the Key Players Driving Innovation?
Major companies and organizations operating in the in vivo CAR-T therapy market include:
- Gilead Sciences (Kite Pharma)
- Novartis AG
- Pfizer Inc.
- Cellectis
- Autolus Therapeutics
- CARsgen Therapeutics
- Bluebird Bio
- Fate Therapeutics
These organizations are investing heavily in advanced delivery systems, AI-assisted therapy optimization, scalable manufacturing platforms, and precision oncology research.
Latest Breakthroughs Reshaping the Industry
Recent innovations are rapidly accelerating momentum in the in vivo CAR-T therapy market:
- Researchers are developing highly sensitive CAR receptors capable of targeting trace proteins in solid tumors, helping overcome major barriers in solid tumor immunotherapy.
- Lipid nanoparticle-mediated mRNA delivery platforms such as GT-801 are enabling direct T-cell programming with streamlined manufacturing capabilities.
- Biotech companies are increasingly focusing on next-generation CAR-T engineering to improve persistence and therapeutic effectiveness in complex tumor environments.
- Industry discussions on Reddit and biotech communities continue highlighting growing investor interest in CAR-T commercialization and next-generation gene-editing platforms.
Challenges: What Could Restrain Market Growth?
Despite extraordinary growth potential, the market still faces several challenges:
- Immunogenicity concerns associated with viral vectors
- Vector stability limitations
- Regulatory complexity surrounding gene therapies
- High research and development costs
- Safety risks related to off-target immune responses
These challenges may slow commercialization timelines and increase development risks for emerging therapy platforms.
Case Study: Simplifying CAR-T Manufacturing Through In Vivo Delivery
Traditional CAR-T therapies often require complex laboratory processing that can take weeks before patient reinfusion. In vivo CAR-T platforms aim to eliminate these bottlenecks by engineering immune cells directly within the patient through intravenous or targeted delivery methods.
This innovation could significantly reduce manufacturing costs, improve accessibility, and shorten treatment timelines, making advanced immunotherapies more scalable for global healthcare systems.
Conclusion: What Lies Ahead for the Market?
The in vivo CAR-T therapy market is poised for explosive growth as biotechnology innovation, AI integration, and advanced gene delivery systems redefine the future of cancer treatment.
With rising clinical trial activity, expanding applications beyond blood cancers, and growing investment from pharmaceutical leaders, in vivo CAR-T therapies are rapidly progressing toward mainstream commercialization. Companies focusing on scalable delivery platforms, precision immunotherapy, and next-generation gene-editing technologies will lead the next era of oncology innovation.
Get Sample Copy: https://www.precedenceresearch.com/sample/8380
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
