The global healthcare industry is entering a transformative phase as regenerative medicine shifts from experimental science toward commercial clinical application. Stem cell therapies are increasingly being explored for the treatment of chronic, degenerative, neurological, cardiovascular, and autoimmune diseases, leading to a sharp rise in large-scale active clinical trials worldwide.
The global large-scale active stem cell clinical trials market was valued at USD 7.20 billion in 2025 and is projected to grow from USD 8.27 billion in 2026 to approximately USD 28.88 billion by 2035, expanding at a CAGR of 14.90% from 2026 to 2035. The market is gaining momentum due to increasing demand for regenerative therapies, expanding clinical trial pipelines, advancements in stem cell engineering technologies, and growing investments from pharmaceutical and biotechnology companies.
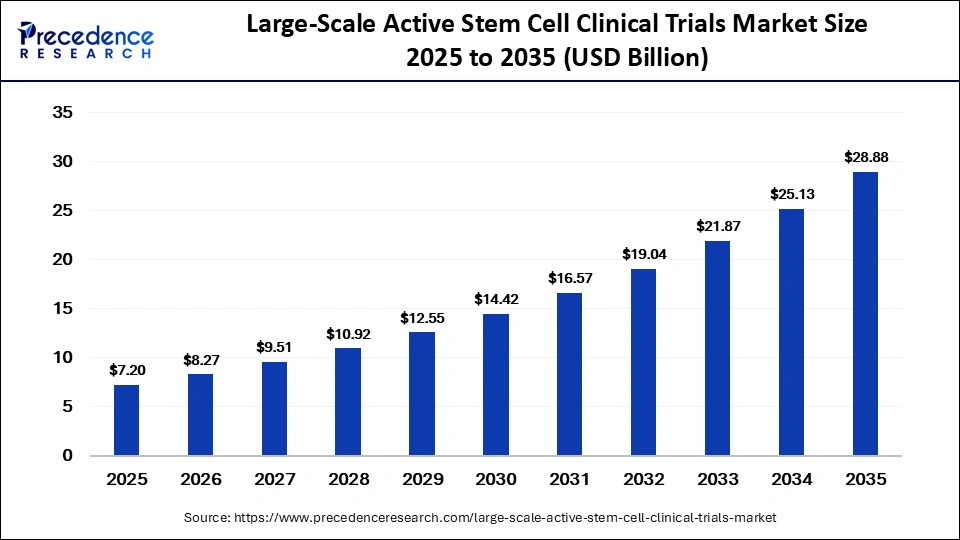
As more therapies move from early-stage studies to advanced Phase III trials, the industry is gradually approaching commercialization and broader clinical adoption. Governments, healthcare institutions, and private investors are increasingly supporting regenerative medicine programs, accelerating global clinical research activity.
Read Also: Oculoplastic Surgery Market
Quick Insights: What Are the Key Market Highlights?
North America dominated the market with a 42% revenue share in 2025 due to strong clinical research infrastructure, advanced biotechnology ecosystems, and high funding availability.
Asia Pacific is expected to grow at the fastest CAGR of 18.5% between 2026 and 2035, driven by rising clinical trial activity and expanding healthcare investments.
Mesenchymal stem cells (MSCs) accounted for the largest stem cell type segment with a 35% market share in 2025.
Induced pluripotent stem cells (iPSCs) are projected to grow at the fastest CAGR of 20.5% due to their versatility and increasing adoption in personalized therapies.
Phase II trials held the largest share of 40% in 2025, reflecting strong mid-stage pipeline activity.
Oncology dominated the application segment with a 35% share due to growing use of stem cell therapies in cancer treatment and immune restoration.
Multi-center trials represented 65% of total market activity as global collaborations become increasingly important for large-scale patient recruitment and regulatory approvals.
Pharmaceutical and biotechnology companies accounted for 50% of total market demand, highlighting growing commercialization efforts.
What Are Large-Scale Active Stem Cell Clinical Trials and Why Are They Important?
Large-scale active stem cell clinical trials are advanced research programs designed to evaluate the safety, efficacy, scalability, and commercialization potential of stem cell therapies across large patient populations. These trials play a critical role in validating regenerative treatments before regulatory approval and widespread medical adoption.
Stem cells possess the unique ability to regenerate damaged tissues and restore biological function, making them highly promising for treating diseases previously considered irreversible. Clinical applications are rapidly expanding across oncology, neurology, cardiovascular disorders, musculoskeletal diseases, and autoimmune conditions.
As global clinical pipelines continue expanding, stem cell therapies are increasingly viewed as one of the most important pillars of future precision medicine.
AI Role in the Market: How Is Artificial Intelligence Accelerating Stem Cell Clinical Trials?
Artificial intelligence is transforming the way stem cell clinical trials are designed, monitored, and optimized. AI-powered analytics platforms help researchers identify ideal patient populations, improve biomarker discovery, and optimize trial protocols for better treatment outcomes.
Machine learning algorithms are also improving stem cell characterization, genomic profiling, and predictive safety analysis. These technologies help reduce trial costs, improve operational efficiency, and accelerate regulatory approvals.
AI-driven monitoring systems additionally enable real-time patient tracking and adverse event prediction during clinical trials, improving both patient safety and study accuracy. As regenerative medicine grows more data-intensive, AI is expected to become central to next-generation stem cell research.
Market Growth Factors: What Is Driving Market Expansion?
Why Is Regenerative Medicine Becoming a Major Healthcare Priority?
Growing prevalence of chronic and degenerative diseases is increasing demand for therapies capable of repairing damaged tissues and restoring biological function. Stem cell therapies are emerging as promising long-term treatment solutions.
How Are Technological Advancements Supporting Market Growth?
Advances in cell engineering, stem cell processing, cryopreservation, and delivery systems are improving therapy scalability and clinical effectiveness.
Why Are Pharmaceutical Companies Increasing Investments?
Pharmaceutical and biotechnology companies are aggressively investing in stem cell pipelines to develop next-generation therapies targeting oncology, autoimmune diseases, and neurological disorders.
How Does Government Support Accelerate Clinical Trials?
Governments and public healthcare agencies are increasing research funding and supporting collaborative clinical trial programs focused on regenerative medicine innovation.
Opportunities & Trends: What’s Shaping the Future of the Market?
Are Induced Pluripotent Stem Cells (iPSCs) Emerging as a Major Innovation Area?
Yes, iPSCs are becoming one of the fastest-growing segments because they can generate multiple cell types while reducing ethical concerns associated with embryonic stem cells.
Can Multi-Center Trials Accelerate Commercialization?
Large-scale multi-center trials are improving recruitment timelines, generating diverse patient data, and strengthening regulatory confidence in stem cell therapies.
Is Oncology Becoming the Largest Therapeutic Application?
Stem cell therapies are increasingly being investigated for cancer treatment, immune reconstitution, and tissue regeneration following chemotherapy.
How Are CROs Transforming Clinical Trial Operations?
Contract Research Organizations (CROs) are becoming essential partners for data management, regulatory compliance, and operational scalability, helping companies accelerate development timelines.
Segmentation Analysis: Where Are the Key Revenue Streams?
By Stem Cell Type
- Mesenchymal stem cells (MSCs): 35% share
- Hematopoietic stem cells (HSCs): strong market presence
- Induced pluripotent stem cells (iPSCs): fastest-growing segment with 20.5% CAGR
- Embryonic stem cells (ESCs): stable growth segment
By Clinical Phase
- Phase II trials: 40% share
- Phase I trials: strong pipeline activity
- Phase III trials: expected CAGR of 17.5% through 2035
By Application
- Oncology: 35% share
- Neurological disorders: fastest-growing application segment
- Cardiovascular diseases
- Orthopedic & musculoskeletal disorders
- Autoimmune diseases
By Therapy Type
- Allogeneic stem cell therapies: 55% share
- Autologous stem cell therapies: rapidly growing personalized treatment segment
By Trial Design
- Multi-center trials: 65% share
- Single-center trials: growing steadily due to personalized treatment approaches
By End-Use
- Pharmaceutical & biotechnology companies: 50% share
- Academic & research institutes: 30% share
- CROs: fastest-growing end-use segment
Regional Analysis: Which Regions Are Leading the Market?
Why Does North America Dominate the Market?
North America leads the global market due to its advanced biotechnology infrastructure, strong regulatory frameworks, robust funding ecosystem, and high concentration of clinical research centers.
How Strong Is Europe’s Position in Stem Cell Research?
Europe holds a significant market share because of strong collaborative research programs, advanced healthcare systems, and supportive regulatory oversight for regenerative medicine studies.
Why Is Asia Pacific the Fastest-Growing Region?
Asia Pacific is witnessing rapid expansion due to growing healthcare investments, large patient populations, government support for biotechnology innovation, and expanding clinical research infrastructure in China, Japan, and India.
Competitive Landscape: Who Are the Key Players Driving Innovation?
Key companies operating in the large-scale active stem cell clinical trials market include:
- Novartis AG
- Fate Therapeutics, Inc.
- Mesoblast Limited
- Athersys, Inc.
- BlueRock Therapeutics
- Vertex Pharmaceuticals Incorporated
- CRISPR Therapeutics AG
- Takeda Pharmaceutical Company Limited
- Lonza Group AG
- Catalent, Inc.
These organizations are investing heavily in scalable manufacturing, cell engineering technologies, AI-driven analytics, and advanced regenerative therapy development.
Get Sample Copy: https://www.precedenceresearch.com/sample/8379
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
